The New Onda
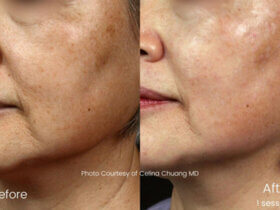
ONDA is the worlds first and only device to use the revolutionary new technology for non-surgical body contouring and fat reduction: Coolwaves™ by DEKA. As manufacturers of the previously globally-renowned Laserlipolysis® treatment, DEKA has again developed a technology that will truly change the way in which all non-surgical body shaping procedures are delivered. The new ONDA device uses unique Coolwaves™ (microwaves) that bypass the uppermost layers of skin tissue, helping deliver more energy/heat directly to where it is needed: the subcutaneous fat cells. This helps dramatically reduce the heating of the surface tissue, resulting in a safer, more effective non-invasive body shaping treatment. Click here to find out more.
Find out more about the Onda, plus the full range of devices available from Lynton Lasers at stand K16 at Aesthetic Medicine Live. Register for your FREE entrance ticket and book CPD-accredited education here.











Leave a Reply