India has enforced new Quality Control Orders (QCOs) for laser-based aesthetic and skin treatment devices. The Department for Promotion of Industry and Internal Trade issued these rules under the Ministry of Commerce and Industry.
Devices must meet Indian standards
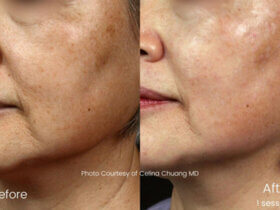
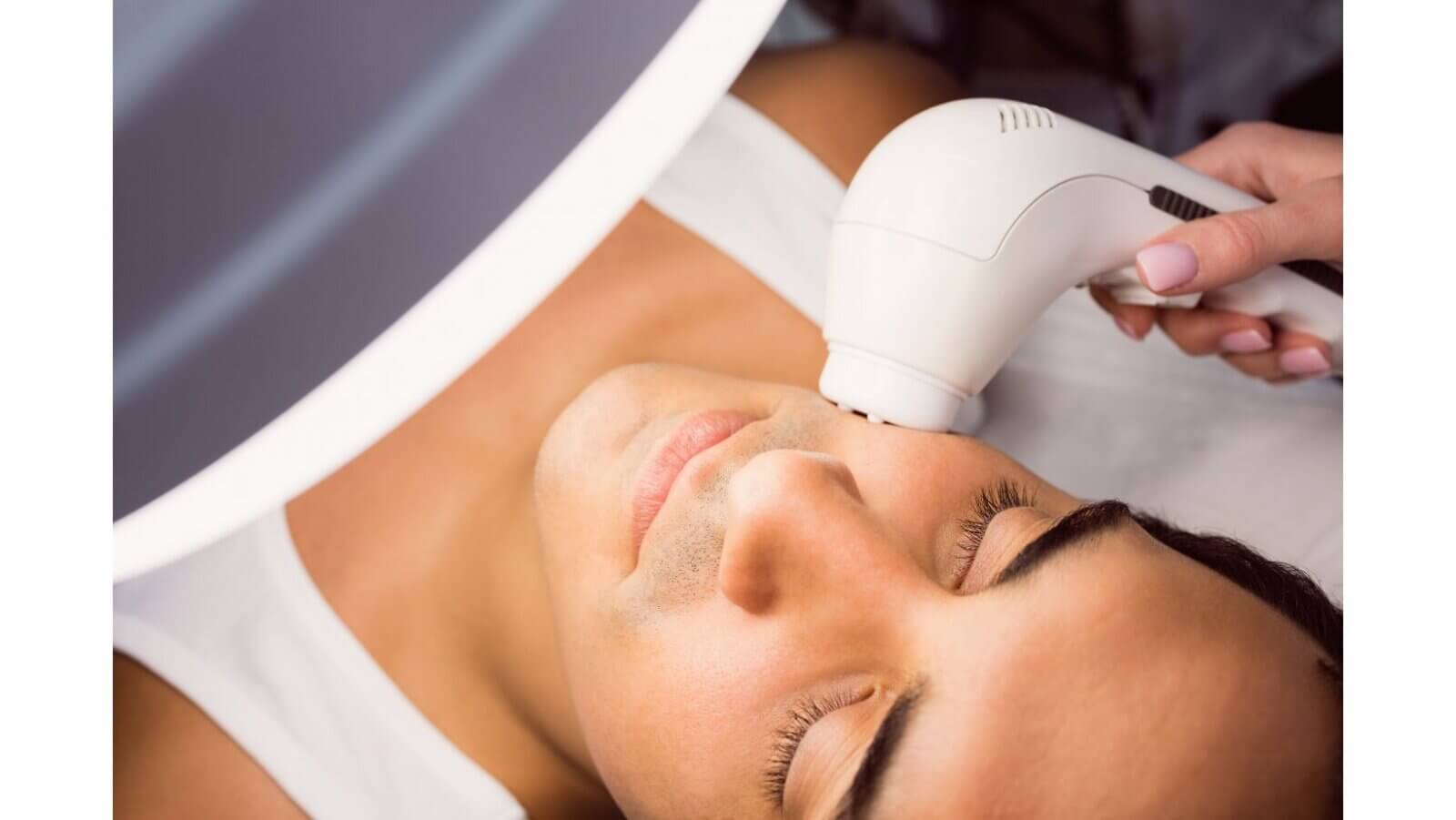
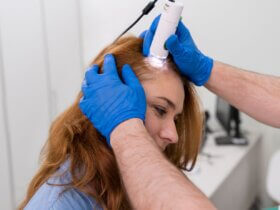
Before manufacturing, importing, or selling in India, devices must meet Indian quality and safety standards. These rules apply to lasers and intense light sources used in beauty and professional aesthetic treatments.
Covered devices include systems for laser hair removal, skin rejuvenation machines, and other electrical optical devices. For electrical safety and reliability, manufacturers and importers must follow Indian Standard IS 302 (Part 1): 2024.
The standard aligns with international norms like IEC 60335-1 to ensure performance and user safety. Companies must obtain a licence from the Bureau of Indian Standards (BIS) to certify compliance before sale.
Non-compliance risks
Both domestic and imported devices must carry a valid BIS Standard Mark under these orders. Products failing to meet standards cannot be sold, stored, or distributed in India legally. Non-compliance may lead to fines, licence suspension, or other penalties under the BIS Act, 2016.
Some devices may also need approval under CDSCO rules depending on intended use claims. Companies must carefully define device usage to determine applicable regulatory requirements and documentation. Industry stakeholders welcome safety standardisation but worry about certification timelines and associated costs.
Compliance deadlines vary by enterprise size, urging companies to start certification early to avoid disruption. BIS approval includes lab testing, documentation review, and Standard Mark labelling on packaging. QCOs reflect India’s broader focus on product safety, consumer protection, and quality assurance.
Market impact
The aesthetic device market is growing, driven by increasing consumer demand for advanced treatments. These rules aim to create a structured quality ecosystem aligned with global safety standards.
Overall, QCO enforcement safeguards users while improving trust in India’s beauty care and aesthetic device sector.