BOTOX® celebrates 30 years of constant development, improvement and study of this medicine and also providing a trusted and effective treatment option for various therapeutic and aesthetic uses
Allergan plc (NYSE: AGN) is commemorating the beginning of the 30th anniversary celebration for BOTOX® with the ringing of the New York Stock Exchange opening bell. Over the past 30 years, more than 100 million vials of BOTOX® and BOTOX® Cosmetic have been manufactured worldwide, providing a trusted and effective treatment option for various therapeutic and aesthetic uses. With two additional U.S. Food and Drug Administration (FDA) approvals this year for therapeutic indications of pediatric upper limb spasticity and pediatric lower limb spasticity, excluding spasticity caused by cerebral palsy, BOTOX® and BOTOX® Cosmetic now proudly hold 14 therapeutic and aesthetic indications combined, reinforcing its leadership in neurotoxin science.
Down the lane
BOTOX® was first FDA-approved in 1989 for two rare eye muscle disorders – blepharospasm and strabismus in adults, making it the world’s first botulinum toxin type A product to be approved. Since then, BOTOX® has continued to innovate which has led to the FDA’s approval of 11 therapeutic indications, including Chronic Migraine, overactive bladder, leakage of urine (incontinence) associated with a neurologic condition, cervical dystonia, adult upper limb spasticity, adult lower limb spasticity, severe underarm sweating (axillary hyperhidrosis), and most recently, pediatric upper limb spasticity and pediatric lower limb spasticity, excluding spasticity caused by cerebral palsy.
Allergan remains committed to pioneering research and development efforts to continue leading innovation in neurotoxins, and to providing robust resources and services to ensure BOTOX® is accessible and affordable to patients, including BOTOX® Savings Program.
BOTOX® Cosmetic
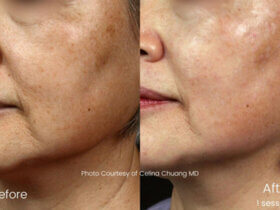
As the first neurotoxin treatment to be approved for aesthetic use, BOTOX® Cosmetic remains a category leader, backed by 17 years of data. Receiving its first FDA approval in 2002 to temporarily improve the appearance of moderate to severe glabellar lines (frown lines between the eyebrows), BOTOX® Cosmetic is also approved for the temporary improvement in the appearance of moderate to severe crow’s feet and forehead lines in adults.
With more than 3,900 articles on BOTOX® and BOTOX® Cosmetic published in scientific and medical journals, and over 115 sponsored studies, BOTOX® neurotoxin is one of the most widely researched medicines in the world. BOTOX® continues to be a leader in its category as it carries 28 indications worldwide, with approval for use in 100 different countries. Backed by strong science and a global presence, BOTOX® proudly embraces its past while boldly looking to the future.
Source: PR News Wire










Leave a Reply