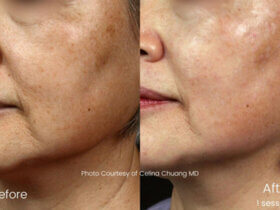
Acrotech Biopharma Inc., the U.S.-based step-down subsidiary of India-headquartered Aurobindo Pharma Ltd., has secured approval from the U.S. Food and Drug Administration for ADQUEY™. The approval marks a significant advancement in the company’s dermatology portfolio. ADQUEY™ is a difamilast 1% ointment for mild-to-moderate atopic dermatitis. The treatment addresses a chronic inflammatory skin condition affecting millions worldwide. The FDA approved the ...
Sensitive skin is now one of the most common skin concerns, affecting nearly 70 per cent of people worldwide. Sensitive skin cases have increased dramatically in 20 years, raising questions about the impact of modern lifestyles and environmental changes. Galderma has been actively studying this trend through its Global Sensitive Skin Faculty, which is a group of leading dermatology experts. ...
Galderma showcased significant updates across its dermatology portfolio at the 2025 American Academy of Dermatology (AAD) Annual Meeting. The company reinforces its leadership commitment in dermatology innovation with 22 scientific presentations, including two oral e-posters. Nemluvio Nemolizumab (Nemulizumab), Galderma’s treatment for prurigo nodularis and atopic dermatitis, was a key focus at AAD 2025. A new oral presentation showcases Nemluvio’s efficacy ...
Dr Mir Shahnawaz, founder of DERMIS, recently discussed advances in treating atopic dermatitis at the RAD Symposium in Jaipur. India’s top dermatologists attended the event, which focused on groundbreaking developments in skin health and innovative treatments. A key highlight was the discussion on Abrocitinib, a JAK1 inhibitor, which has shown promising results for treating moderate to severe atopic dermatitis. Atopic ...